Overview
Research in our lab aims to understand the mechanism and regulation of homologous recombination during meiosis, and the influence of genetics and the environment on this essential process. Meiotic recombination is essential for accurate chromosome segregation at the first meiotic division, and produces new combinations of gene alleles upon which natural selection can act. Infertility, pregnancy miscarriage, aneuploid diseases (such as Down Syndrome) and de novo chromosome rearrangements are associated with defects in meiotic recombination, making it the leading cause of congenital disease. In recent years, my lab has expanded from its core of yeast molecular genetics, in order to pursue a more integrated approach to understanding meiotic recombination, with new focuses on mammalian meiosis and the regulation of recombination by post-translational protein modification.
Early in meiosis, hundreds of programmed DNA breaks are inflicted to initiate the templated repair process called homologous recombination. Homologous recombination facilitates chromosome pairing during meiosis and promotes their accurate segregation at the first meiotic division. Recombination is carefully regulated such that each pair of chromosomes always becomes connected by at least one crossover, which is required for stable bi-orientation on the meiosis I spindle.

+ Regulation of meiotic prophase by post-translation modification
RNF212, the first factor to be implicated in heritable variation in human crossover rate, is essential for crossing over in mouse, but remains proficient for the repair of the hundreds of meiotically-induced DNA double-strand breaks. We further showed that the RNF212 protein selectively localizes to a subset of DNA double-strand breaks, where it stabilizes factors required for crossing over. Given that RNF212 is an E3-ligase for SUMO conjugation, this study established a new paradigm for understanding how meiotic cells establish crossover sites.
+ Formation and resolution of DNA joint molecules during homologous recombination
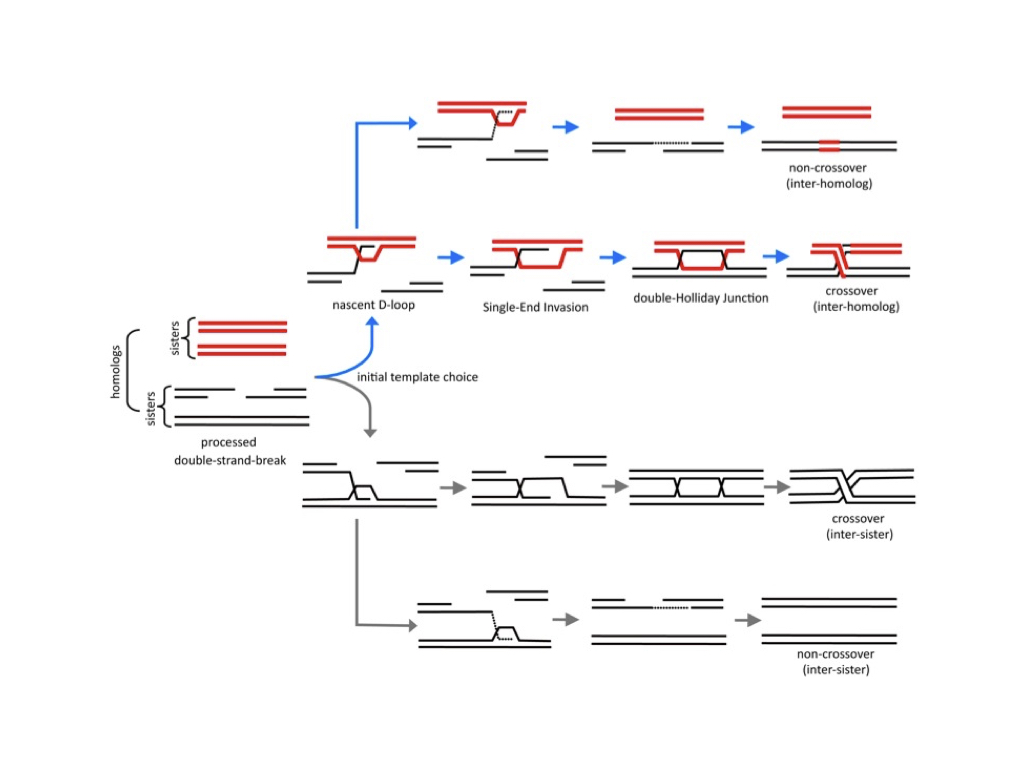
Homologous recombination occurs when a broken chromosome uses a homologous chromosome as a template for its repair. The central reaction of homologous recombination is the formation of a Joint Molecule intermediate via pairing and strand-exchange between a free DNA end and a homologous template chromosome.
Two basic types of Joint Molecule may form depending on whether one or both ends of a broken chromosome engage the template. A D-loop is formed when one DNA end undergoes strand-exchange, and a double Holliday Junction forms when the second end is also engaged. After a Joint Molecule has formed sequences can be copied from the template chromosome via DNA synthesis. Finally, the Joint Molecule must be resolved or dissociated. Resolution can occur with one of two outcomes: a crossover, with exchange of chromosome arms, or a non-crossover involving only a local alteration of DNA. Our research focuses on understanding how the formation and resolution of Joint Molecules is regulated and coordinated with other cellular events.
During meiotic prophase, recombination plays critical roles for the pairing and segregation of parental chromosomes (homologs). In particular, crossing-over between homologs facilitates their stable bipolar connection to the meiosis I spindle and thereby promotes accurate homolog disjunction. Meiotic cells tightly regulate hundreds of recombination events so that at least one crossover (but not many) is formed between each homolog pair. Meiotic recombination is regulated at four distinct levels: (1) Template Choice – homologs are utilized preferentially over sister-chromatids; (2) Joint Molecule Formation – only crossover-designated events form stable double Holliday Junctions; (3) Joint Molecule Resolution – double Holliday Junction resolution must be biased to produce a crossover outcome and D-loops must be completely dissociated to restore duplex continuity and permit chromosome segregation; (4) Coordination with Chromosome Morphogenesis – the progression of recombination must be coordinated with homolog pairing, synapsis and segregation. Defective meiotic recombination is linked to infertility and aneuploid diseases such as Down’s Syndrome.
In somatic cells, unregulated crossing-over can cause chromosome rearrangement and missegregation and the homozygosis of deleterious mutations. To minimize these risks, mitotically cycling cells actively